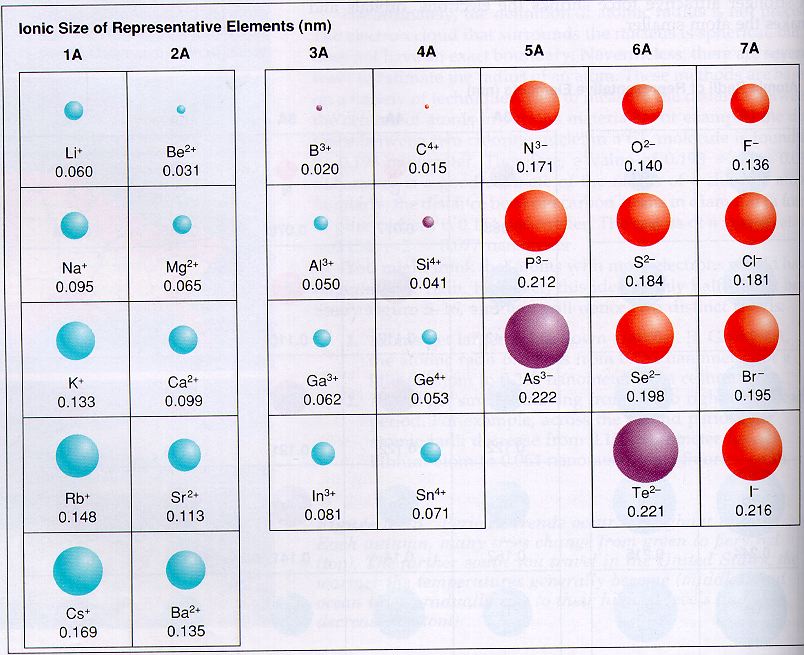
It also depends on the spin state of the ion. It depends on the coordination number, or the number of atoms, ions, or molecules that a central atom or ion holds as its nearest neighbors in a complex or coordination compound. Moreover, the ionic radius is tricky to measure since it depends on the varying factors of the environment in which the ion is located. Since ions tend to exist in bonds, the ionic radius can be found via the ionic bond between two atoms. The ionic radius can easily be a little smaller or larger than the atomic radius, which is the radius a neutral atom of the element possesses. So the ions of an atom are treated as if they were spheres. As mentioned above, the shell boundaries of electron shells are difficult to read. The ionic radius is half of the distance between two ions that is just touching one another. The ionic radius of an ion is measured when the atom is in a crystal lattice structure. “How to Find the Number of Valence Electrons?” Science ABC, 17 Jan. This is a good visualization of how the radius may be measured but this is unlikely to be accurate because the borders of orbitals are quite fuzzy. To the red electrons at the outermost orbital. So in the picture below the ionic radius would be found by measuring the distance between the yellow and blue protons in the nucleus. Ionic Radiusīy definition, an ionic radius is the distance of the outermost shell of electrons (valence shell) from the nucleus of an ion. For example, the metals in group 1A (Alkali Metals) all have a +1 charge meaning they want to give away an electron. Most metals on the periodic table tend to form cations. In contrast, when an atom in the periodic table gains an electron it becomes an anion. When an atom on the periodic table loses an electron it becomes a cation. Valence Electrons– the electrons in the outermost shell, or energy level, of an atom.Īn ion is an atom that carries a positive or negative electric charge.Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice. Ionic Radius – is the radius of a monatomic ion in an ionic crystal structure.Atomic Radius – The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron.You will also be introduced to the concepts that contribute to ionic radius, including how to find it. Use Internuclear distance formula to find the r +.įigure 3: The Hard sphere model can roughly determine the ion radii.In this tutorial, you will be introduced to ionic radius trends on the periodic table of elements.Find internuclear distance (d) between anion and cation.This will minize the distribution of electrons. *To calculate ion radii, Lande used ionic compound under solid state (ex: NaCl). Internuclear distance (d) = r cation + r anion 2 In general, scientists uses formula of Internuclear distance to test out the radii of ion then compared with the ion radii had done on X-ray diffraction: Figure 2: 3-D hard sphere model of Sodium Chloride, taken with permission from en./wiki/File:So.e-3D-ionic.png The Hard-Sphere model can be applied to metallic and ionic compounds such as NaCl, which is shown below. Figure 1: Schematic of the hard-sphere model 
The ion radii measured under crystal state of ionic compound which cations and anions are stacking in pattern as shown below. 5 The Hard-Sphere model has been tested by well-known scientists Lande', Pauling and Goldsmidt. The Hard-Sphere model are impenetrable spheres that do not overlap in space.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
